Vaccine Development CRO Services
As an expert in providing solutions to assist virology and microbiology research, Creative Diagnostics is proud to introduce comprehensive Vaccine Development CRO Services to support the development of innovative vaccines for infectious diseases. With expertise spanning a range of integrated technology platforms, Creative Diagnostics provides custom solutions for various preclinical vaccine development needs, such as antigen discovery, adjuvant selection, immunogenicity testing, and efficacy testing.
Vaccine development is a very complex process involving multiple stages from basic science through clinical trials to industrial production and commercialization. Vaccine research and development is a multidisciplinary endeavour that requires significant resources. As science and technology have evolved, new technologies have made it possible to keep vaccine development on schedule and ensure the safety and efficacy of vaccines. At the same time, global collaboration is an important driver of vaccine development, especially during global epidemics.
Creative Diagnostics offers comprehensive and customized preclinical CRO services to support vaccine development, e.g., antigen discovery and validation, adjuvant selection and biomarker development. First, when a project is initiated, Creative Diagnostics conducts a thorough assessment based on the client's specific needs. The company's team of experts then works together to implement the project with strict quality control and timely updates. Finally, Creative Diagnostics provides a complete final report with an emphasis on openness, efficacy and compliance to ensure that the client's vaccine candidate is approved for marketing.
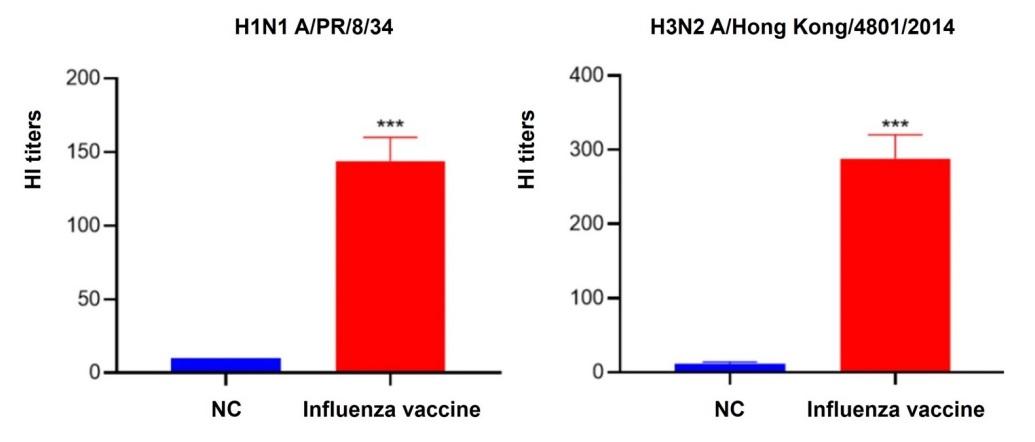
Creative Diagnostics can help clients navigate the complex landscape of preclinical vaccine research, from identifying target antigens and selecting appropriate animal models, to optimizing vaccine formulations and conducting rigorous safety and efficacy assessments. For instance, Creative Diagnostics offers various virus/antibody-based assays to test vaccine immunogenicity to ensure that the vaccine candidate can generate sufficient antibody and/or cellular immune responses to provide protection.
In addition, Creative Diagnostics offers a wide range of testing options to ensure accurate evaluation of vaccines, enabling clients to gain insight and improve vaccine design and production efficiencies to ensure vaccine safety and efficacy. These services include in vitro and in vivo relative potency assays (antigen detection, ED50 values, neutralizing antibody detection), cytokine panels, ELISA, flow cytometry, in vivo small animal imaging, histopathology, immunofluorescence, western blotting, and ELISPOT assays.
The company also provides a wide range of neutralizing antibodies against infectious disease pathogens. These antibodies can be used in a variety of applications including ELISA kits, neutralization assays, WB, IHC and other procedures. The host switching and antibody modification services are also available on request, as well as pseudotyped reporter viruses and virus-derived peptides against multiple targets.
Creative Diagnostics has extensive experience in developing platforms for live attenuated, inactivated, conjugate, recombinant protein, nucleic acid and viral vector vaccines. With a full range of capabilities, Creative Diagnostics is a trusted CRO service provider that delivers innovative, tailored solutions to accelerate vaccine innovation and successfully prevent infectious diseases. For more information on Creative Diagnostics' Vaccine Development services and other innovative solutions, please visit https://antiviral.creative-diagnostics.com/vaccine-development.html.
About Creative Diagnostics
Headquartered in New York, Creative Diagnostics is a consulting and experimental service provider specializing in virology and microbiology. The company provides comprehensive solutions to conquer obstacles in virology and microbiology research, from high-security infrastructure provision, biosafety regulation elucidation, to expert viral system assistance.
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Παιχνίδια
- Gardening
- Health
- Κεντρική Σελίδα
- Literature
- Music
- Networking
- άλλο
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness
